Transcranial Magnetic Stimulation in Addiction Treatment: A Consultant Addiction Psychiatrist’s Perspective
By Dr. Sharad Haridas, MBBS, MRCPsych (London), FAMS (Psych)

Dr. Sharad Haridas, MBBS, MRCPsych (London), FAMS (Psych)
Dr. Haridas is a dual-registered consultant psychiatrist and addiction specialist with 16 years of experience in London. At the Winslow, he oversees TMS for clients with addiction using evidence-based, personalised treatment pathways. He is a Fellow of the Clinical TMS society (International).
For those working at the intersection of addiction psychiatry and neuromodulation, it is both an exciting and a cautiously optimistic time. Transcranial magnetic stimulation (TMS) has been a tool in the psychiatric armamentarium for four decades now, since Professor Anthony Barker and his team at the University of Sheffield demonstrated the first functional TMS device in 1985. In that time, the technology has matured from a laboratory curiosity into a clinically validated treatment for depression, obsessive-compulsive disorder, migraine, and most recently, smoking cessation. Its application to substance use disorders is still developing. It shows real promise, but research methods and heterogeneity vary widely, and institutions seem to be rightly but excessively cautious about adopting it as a standard treatment.
This article is not intended as an exhaustive systematic review. Rather, it reflects the perspective of a clinician who has been delivering TMS in the context of addiction treatment for a number of years, and who wishes to share a practical, evidence-informed account of where TMS fits — and where it does not — in the treatment of substance use disorders.
A Primer on the Mechanism
For colleagues less familiar with TMS, the basic mechanism is worth revisiting. A figure-of-eight coil is placed against the scalp. An electrical current passes through the coil, which in turn generates a focused magnetic field that passes through the skull without impedance. This then induces a local electrical current in the underlying cortical tissue causing depolarisation. Unlike electroconvulsive therapy (ECT), which delivers electrical current directly to the scalp to induce a generalised seizure, TMS is typically administered at subthreshold levels for seizure induction while remaining above the threshold for local depolarization. The individual remains fully conscious throughout, experiences no anaesthesia, and can typically resume normal activities immediately after treatment.
The most common target in both depression and addiction protocols is the left dorsolateral prefrontal cortex (DLPFC), a region central to executive function, impulse control, and top-down regulation of deeper limbic and reward-related brain structures. In addiction, there is compelling evidence that the prefrontal cortex is functionally underactive, particularly in alcohol and stimulant use disorders, while subcortical structures — the amygdala, insula, and ventral striatum — tend toward overactivation. This mismatch between cortical control and subcortical drive is a hallmark of the addicted brain, and it maps closely onto what is observed in addiction or any compulsive use behaviours: diminished impulse control, heightened cue reactivity, and difficulty maintaining behavioural change despite sincere motivation.
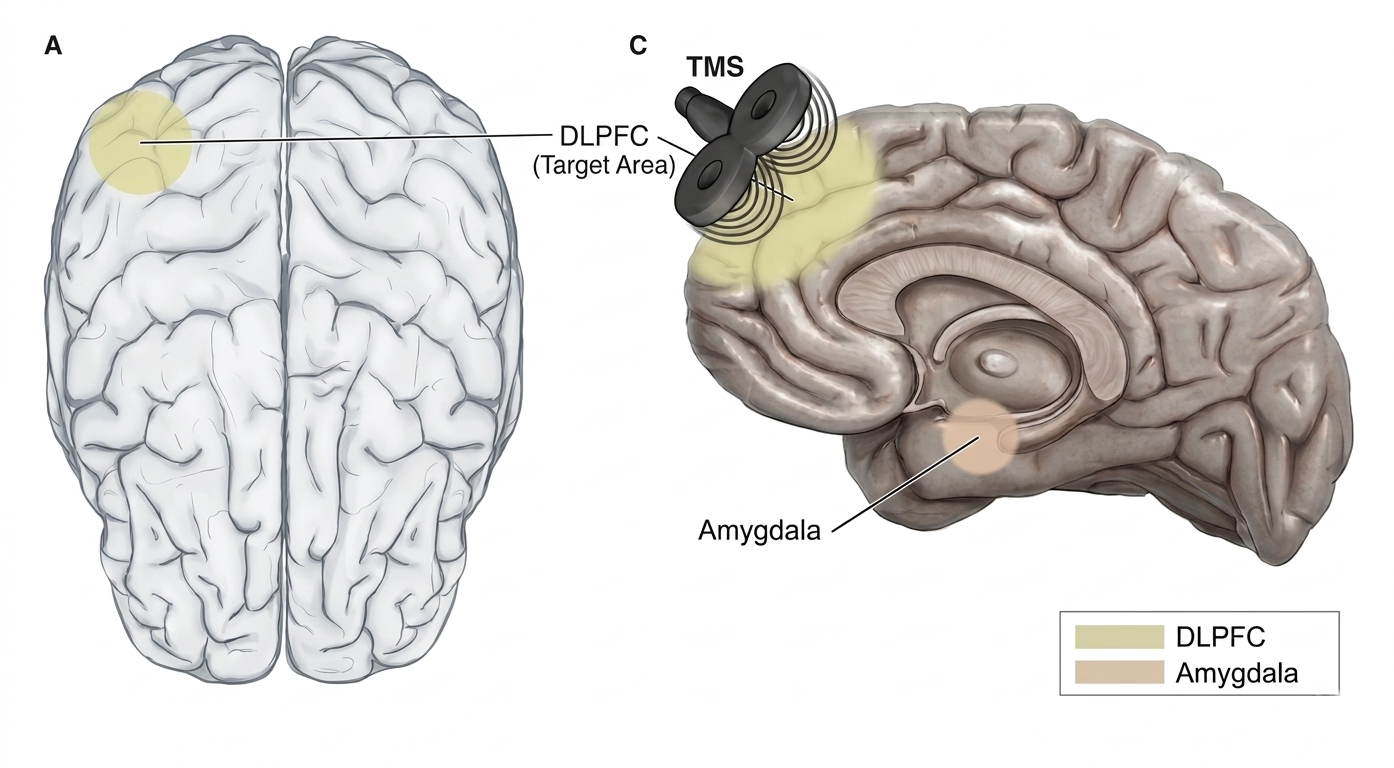
The rationale for TMS in addiction is therefore relatively intuitive: by upregulating prefrontal activity through high-frequency stimulation (typically 10 Hz, or via intermittent theta burst protocols), we can potentially strengthen the cortical circuits responsible for inhibitory control and cognitive flexibility, while indirectly modulating the hyperactive subcortical networks that drive craving and compulsive use. There is also evidence that repetitive TMS promotes dopamine release in the mesolimbic and mesocortical pathways, which may help to partially restore the blunted reward processing that characterises chronic substance use and impulsive control behaviours.
The Evidence Base: Honest and Transparent
It is important to be candid about where the evidence currently stands. The body of research on TMS for substance use disorders has grown substantially, and especially in the last decade. A 2025 analysis identified 190 published articles on TMS-based addiction treatment spanning 2001 to 2023, with clinical studies representing the largest category. Systematic reviews and meta-analyses have consistently reported that repetitive TMS targeting the DLPFC produces a statistically significant reduction in craving across multiple substance types, with pooled effect sizes falling in the moderate range. Randomised controlled trials have demonstrated reductions in craving and use across tobacco, alcohol, cocaine, methamphetamine, opioid, and cannabis use disorders.
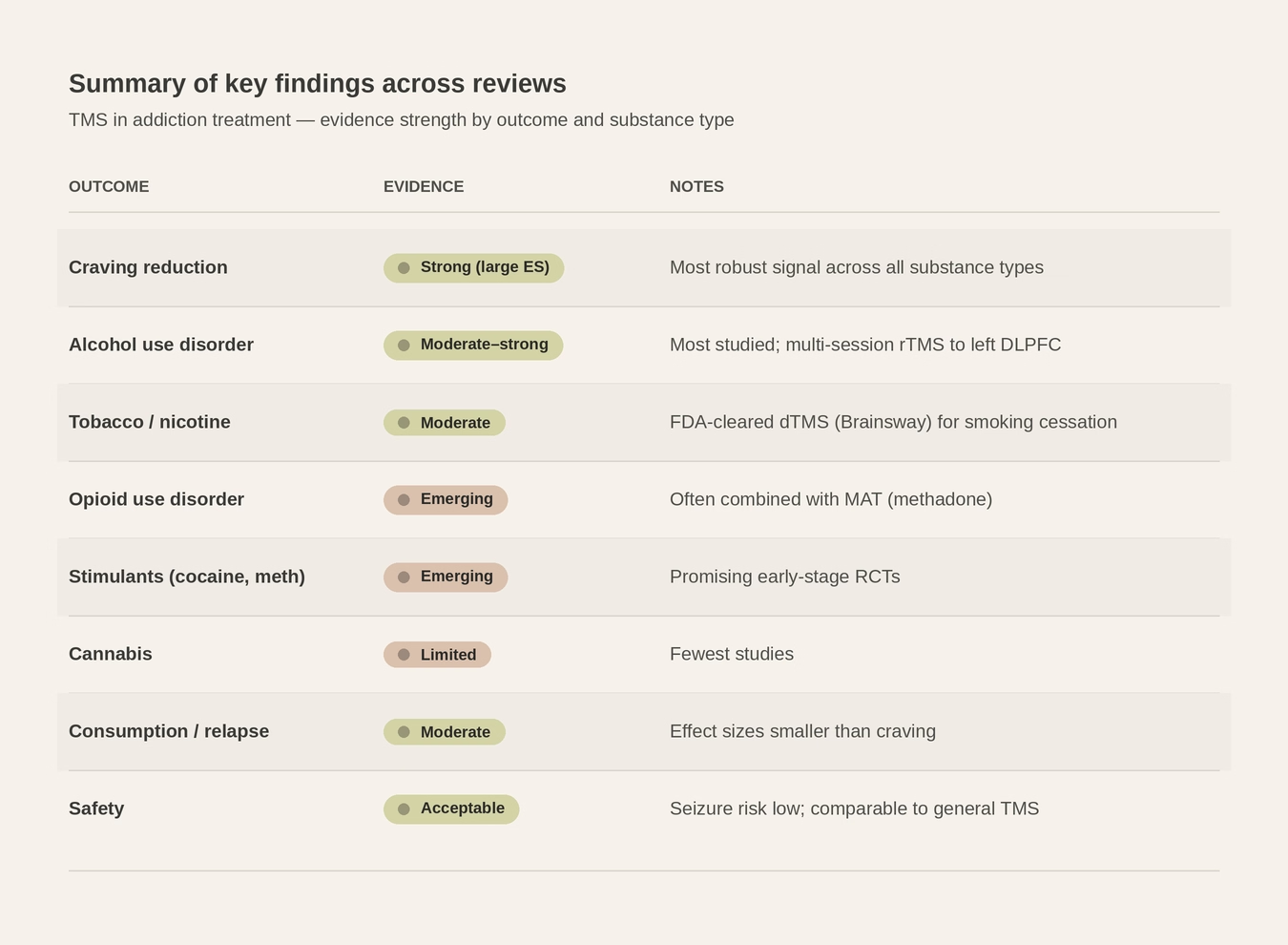
The effect size for substance use outcomes is moderate. Whilst it may not rival the magnitude of effect we see with TMS for major depressive disorder, where the treatment has a deeper and more robust evidence base, it is posited to have a moderate effect size in the context of addiction, where relapse rates with conventional treatment remain discouragingly high. It is therefore not something to dismiss. When TMS is framed as an adjunctive treatment to psychotherapy and pharmacological management, the clinical value becomes much clearer. This distinction is essential when communicating with patients, families, and referrers.
FDA-Cleared Indications: Where We Stand
The regulatory trajectory of TMS has been a slow but steady progression of approved indications.
2008
Major depressive disorder – high-frequency left DLPFC stimulation.
2013
Migraine – single-pulse TMS for acute treatment of migraine with aura.
2018
Obsessive-compulsive disorder – BrainsWay Deep TMS targeting medial prefrontal and anterior cingulate cortices.
2020
Smoking cessation – BrainsWay Deep TMS with H4 coil targeting bilateral insula and prefrontal cortices; the first FDA clearance for any TMS device in the addiction space.
The smoking cessation clearance was a landmark moment. The pivotal trial involved 262 adults with long histories of heavy smoking and repeated failed quit attempts. Among those who completed the full course, the quit rate was 28.4% in active TMS versus 11.7% in sham — a statistically significant and clinically meaningful difference. No seizures were observed; the most common side effects were headache and application site discomfort.
In parallel to the FDA developments, MagVenture TMS Therapy® received CE approval in April 2021 for the treatment of psychoactive substance use disorder (PSUD) in adults — representing the world’s first CE approval for TMS in addiction treatment. This approval, based on a naturalistic study demonstrating 69% effectiveness versus 19% for standard care, marked a significant regulatory milestone in Europe and positioned TMS as a legitimate treatment option for a broader range of substance use disorders beyond tobacco.
At present, TMS holds specific FDA clearance for nicotine addiction. This should not be confused with a lack of evidence or efficacy for other substances/addictions. The regulatory process is slow, expensive, and heavily influenced by commercial viability. The absence of a formal indication does not mean that TMS lacks utility in these contexts, it means that the pathway to formal approval has not yet been completed. In the meantime, a growing number of clinicians worldwide are delivering TMS for alcohol, stimulant, and other substance use disorders on the basis of the existing clinical literature and expert consensus.
Substance-Specific Considerations
In clinical practice, the strength of the evidence and the magnitude of benefit vary by substance. Based on the available literature and our own clinical experience, the following general patterns emerge.
Tobacco
This is where the evidence is strongest and most mature. The BrainsWay H4 coil protocol for smoking cessation is the only FDA-cleared TMS application in the addiction domain. Beyond the pivotal trial, numerous smaller studies directed at the DLPFC have shown reductions in cigarette consumption and craving.
Stimulants (Cocaine and Methamphetamine)
The data here is encouraging. One of the primary brain circuits implicated in cocaine and methamphetamine use disorders is the mesolimbic reward pathway. Both substances increase dopamine transmission throughout the mesolimbic system, which is implicated in drug-seeking and relapse.
Repeated stimulant exposure, with its associated surges in dopaminergic transmission, is thought to induce longer‑term changes in glutamatergic signalling within the prefrontal cortex, thereby exacerbating the imbalance between executive control (inhibitory) and reward networks (drug‑seeking drives). This network dysregulation is associated with impaired control, heightened cravings, and the maintenance of stimulant addiction.
Several open-label and controlled trials have shown reductions in craving and use. A large multi-site RCT (STIMULUS) should substantially clarify the efficacy of TMS for stimulant use disorders in the coming years.
Alcohol
The evidence for alcohol use disorder is moderate. Several trials have demonstrated reductions in craving with high-frequency left DLPFC stimulation, though results are somewhat inconsistent across studies, likely owing to higher heterogeneity. Emerging approaches targeting the salience network, and in particular the insular cortex, may prove more fruitful for alcohol use disorder specifically. TMS does still reduce cravings for alcohol, and in those who have an underlying depressive disorder, it may prove to be even more beneficial.
Opioids and Cannabis
The evidence base for these substances is thinner but growing. Early-stage trials suggest possible benefits, and ongoing research is exploring both conventional rTMS and theta burst protocols. These are areas to watch.
Practical Considerations
Delivering TMS within a residential rehabilitation setting has several advantages. Firstly, clients are in a structured, supervised environment. Delivering twice-daily treatment sessions is straightforward when the individual is not navigating the demands of daily life, work and triggers that drive their substance/behavioural use. TMS can be seamlessly integrated alongside psychotherapy, group work, and other therapeutic activities without disrupting the overall treatment programme.
In our clinical practice, we have found it most effective to start TMS in the second week of admission. The first week is usually focused on settling in, medical stabilisation, and managing acute withdrawal. Initiating TMS after this initial phase—when the individual is physiologically stable and more psychologically receptive — appears to optimise both engagement and tolerability.
We typically administer twice-daily sessions, five days per week, aiming for ~ 30-36 sessions. This aligns with the evidence suggesting that 30 sessions (up to 36) is likely the threshold for the maximum therapeutic benefit. Emerging accelerated protocols — notably iTBS — have compressed session times dramatically. Where a conventional 10 Hz rTMS session takes approximately 37 min, an iTBS session can be delivered in 3-4 min. This approach, pioneered in the Stanford Neuromodulation Therapy (SAINT) protocol, is now being explored for addiction applications, with early proof-of-concept studies in cocaine use disorder demonstrating both safety and tolerability.
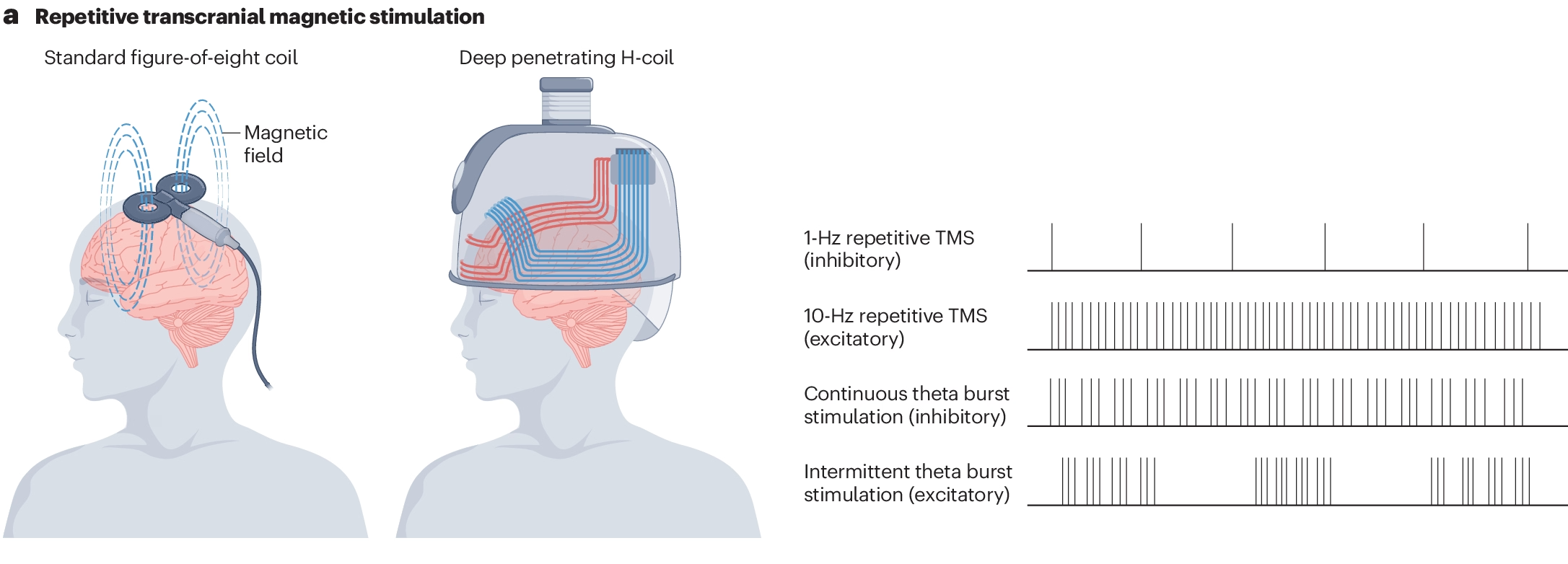
Source: Nature Reviews Psychology https://nature.com/articles/s44159-024-00291-3
Sessions are spaced 50 minutes apart, consistent with the protocols in accelerated TMS trials. This interval is thought to be necessary for the consolidation of the neuroplastic changes induced by each stimulation session.
Client Selection and Safety Screening
Not everyone entering rehabilitation is a suitable candidate for TMS, and careful screening is essential. The ideal candidate, in our experience, is someone presenting with a substance use disorder alongside a comorbid psychiatric condition such as depression, anxiety, OCD, or PTSD. For these individuals, TMS offers the possibility of addressing both the addiction and the underlying psychiatric condition simultaneously, a particularly appealing proposition given how frequently these conditions are intertwined. The individual whose depression drives compulsive substance use, or whose PTSD-related hyperarousal perpetuates reliance on alcohol or benzodiazepines to self-medicate, potentially stands to benefit the most from TMS as an adjunctive treatment.
People in the acute stages of stimulant withdrawal (particularly from high doses), those whose medications have just been significantly altered, and on medications that substantially lower the seizure threshold will generally be considered less suitable for TMS. Individuals with a pre-existing seizure disorder or significant risk factors for seizures are generally excluded. It is important to put this risk in context: large surveys suggest that seizures during TMS are rare, with rates well below 1 per 1000, when treatments are delivered within safety guidelines and obvious risk factors are excluded.
We provide all prospective clients with comprehensive information about TMS, including a summary of the research evidence, its strengths and its limitations, and a frank discussion of what they can realistically expect. TMS will be offered on an entirely optional basis, and informed consent is central to the process.
Side Effects and Tolerability
One of the most compelling features of TMS, particularly in comparison to standard drug treatments and certainly to ECT, is its favourable side-effect profile. The most commonly reported adverse effects are localised headache at the stimulation site and, in some cases, initial insomnia. Both are typically mild and self-limiting, and can be managed with standard analgesics and dose adjustment respectively. Most clients tolerate the treatment well, and dropout rates in clinical trials have generally been low.
It is worth explicitly addressing the frequent conflation of TMS with ECT, which many still make. The two treatments are fundamentally different. ECT induces a generalised seizure under general anaesthesia, with well-documented cognitive side effects. TMS does none of these things. There is no anaesthesia, no seizure, and no alteration in the level of consciousness. Clients can watch television, listen to music, or simply relax during treatment. The sensation is typically described as a light tapping on the scalp — nothing more.
Why TMS Remains Underutilised
Despite four decades of research and a steadily expanding evidence base, TMS remains markedly underutilised in addiction treatment settings worldwide. The reasons for this are multifactorial and worth examining.
First, there is the inherent conservatism of the medical profession. New treatments, even those with reasonable evidence, take time to translate to clinical practice. TMS for depression took over a decade from the initial positive trials to widespread adoption, and the trajectory for addiction applications is following a similar pattern.
Second, there are cost and infrastructure barriers. TMS machines are not inexpensive, and delivery requires trained staff, appropriate clinical space, and ongoing quality assurance.
Third, many addiction treatment services, particularly those in the public sector, are already stretched thin and may not have the capacity to introduce a new treatment modality.
Finally, there is a legitimate concern about the strength of the evidence — the effect sizes are moderate, the protocols are not yet standardised, and many of the published studies are small or methodologically heterogeneous.
These are all valid considerations. I would argue however that the evidence for TMS as an adjunctive treatment for substance use disorders is at least as strong as — and in many cases stronger than — the evidence for routine treatment as usual offered in rehabilitation settings. In a field that continues to struggle with high relapse rates despite treatment, adding a safe, well-tolerated, non-pharmacological modality seems not only reasonable but prudent.
Ongoing Treatment and Relapse Prevention
The therapeutic effects of TMS are not always immediately apparent. While some report improvements within the first ten sessions, the majority begin to notice benefits in the latter stages and, more typically, four to six weeks afterwards. This delayed onset is consistent with the neuroplastic mechanisms thought to underlie TMS’s effects and should be communicated clearly at the outset, to avoid premature disengagement due to unrealistic expectations.
For those who experience relapse following an initial successful course of TMS, maintenance or “top-up” sessions at a reduced frequency can be useful. This is analogous to the maintenance approaches for TMS for depression, where periodic booster sessions have been shown to help sustain remission. The optimal frequency and duration of maintenance TMS for addiction have not yet been formally established, but clinical experience suggests that brief courses delivered at the earliest signs of relapse or increased craving can be beneficial.
Appeal of a Non-Pharmacological Option
A subset of those presenting for addiction treatment have never been on psychiatric medication and are actively reluctant to start. For these individuals, TMS should be viewed as a particularly attractive option: a treatment that targets the neural circuitry underlying their condition without requiring them to take a daily medication.
Moving Forward
The clinical practice of neuromodulation for addiction is evolving rapidly. Several developments are likely to shape clinical practice in the coming years. Large-scale double blind RCTs like STIMULUS for stimulant use disorders (cocaine and methamphetamine) will provide the kind of robust evidence needed to move the field from promising to established. The trial is primarily a feasibility and safety test of delivering high frequency TMS in this population, but it is also powered to give early signals on abstinence and craving.
Accelerated theta burst protocols (like iTBS), may eventually compress the usual 30-session treatment course into days rather than weeks, making TMS accessible to people in short-stay or outpatient contexts.
TMS has great potential as a novel intervention for SUDs. As an adjunct offered alongside treatment as usual, it clearly represents one of the most promising additions to the addiction treatment landscape in recent years. For colleagues considering the introduction of TMS into clinical practice, I would offer this encouragement: the evidence supports its use, the safety profile is reassuring, people tolerate it well, and the potential to address both addiction and comorbid psychiatric conditions with a single modality is a distinct clinical advantage. The conservative instincts that have slowed the uptake of TMS are understandable, and are aligned with the conservative nature of medicine and psychiatry in general, and it is incumbent upon clinicians to better educate themselves on how to begin configuring, delivering and integrating TMS within existing care pathways.